There are many ways to produce salt. Like we talked about in the neutralization reaction the following produce salt:
Acid + Base = Salt + H2O
Acid +Alkali = Salt + H2O
Acid + Carbonate = Salt + H2O + Carbon Dioxide
Acid + Metal = Hydrogen Gas (H2)
Seem a little repetitive? GOOD!! thats how you will remember it.
- Acid + Alkali
- Acid + Base
- Acid + Carbonate
- Acid + Metal
Acid + Base = Salt + H2O
Acid +Alkali = Salt + H2O
Acid + Carbonate = Salt + H2O + Carbon Dioxide
Acid + Metal = Hydrogen Gas (H2)
Seem a little repetitive? GOOD!! thats how you will remember it.
this page will show you the process of making salts with and acid and base
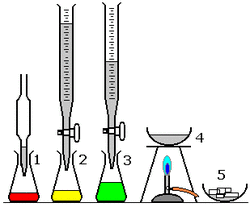
Procedure
1. Put on goggles, tie back hair and make sure you have no electronics near where the experiment is being preformed
2. Take 1 200 cc flask and measure its weight in grams
3. Then take out 2 100cc beakers, a hot plate and two-pipette pump.
4. Pour 40 cc of Sodium hydroxide in one beaker and 40cc of Hydrochloric Acid in another beaker and label them.
5. Use one of the pipette pumps and place it into the Sodium Hydroxide and measure 10cc with it.
6. Put the 10cc of Sodium Hydroxide and put it into the flask
7. Then use the other pipette pump and place it into the Hydrochloric Acid and measure 10cc with it.
8. Put the 10cc of Hydrochloric Acid and put it into the flask.
9. Spin the flask around softly to stir the Hydrochloric Acid and Sodium Hydroxide together
10. Now set the hot plate to the highest heat setting and place the flask onto it.
11. Leave the flask onto the hotplate until all of the H20 has evaporated and only NaCl is left over in the flask.
12. Remove the flask using tongs so you do not burn yourself
13. Take the final weight of the flask with NaCl (Salt) in it and subtract it by the initial weight of the flask.
14. Record all the observations and results from this experiment.
15. Clean all equipment and put it back into the closets in the chemistry room.
1. Put on goggles, tie back hair and make sure you have no electronics near where the experiment is being preformed
2. Take 1 200 cc flask and measure its weight in grams
3. Then take out 2 100cc beakers, a hot plate and two-pipette pump.
4. Pour 40 cc of Sodium hydroxide in one beaker and 40cc of Hydrochloric Acid in another beaker and label them.
5. Use one of the pipette pumps and place it into the Sodium Hydroxide and measure 10cc with it.
6. Put the 10cc of Sodium Hydroxide and put it into the flask
7. Then use the other pipette pump and place it into the Hydrochloric Acid and measure 10cc with it.
8. Put the 10cc of Hydrochloric Acid and put it into the flask.
9. Spin the flask around softly to stir the Hydrochloric Acid and Sodium Hydroxide together
10. Now set the hot plate to the highest heat setting and place the flask onto it.
11. Leave the flask onto the hotplate until all of the H20 has evaporated and only NaCl is left over in the flask.
12. Remove the flask using tongs so you do not burn yourself
13. Take the final weight of the flask with NaCl (Salt) in it and subtract it by the initial weight of the flask.
14. Record all the observations and results from this experiment.
15. Clean all equipment and put it back into the closets in the chemistry room.
